
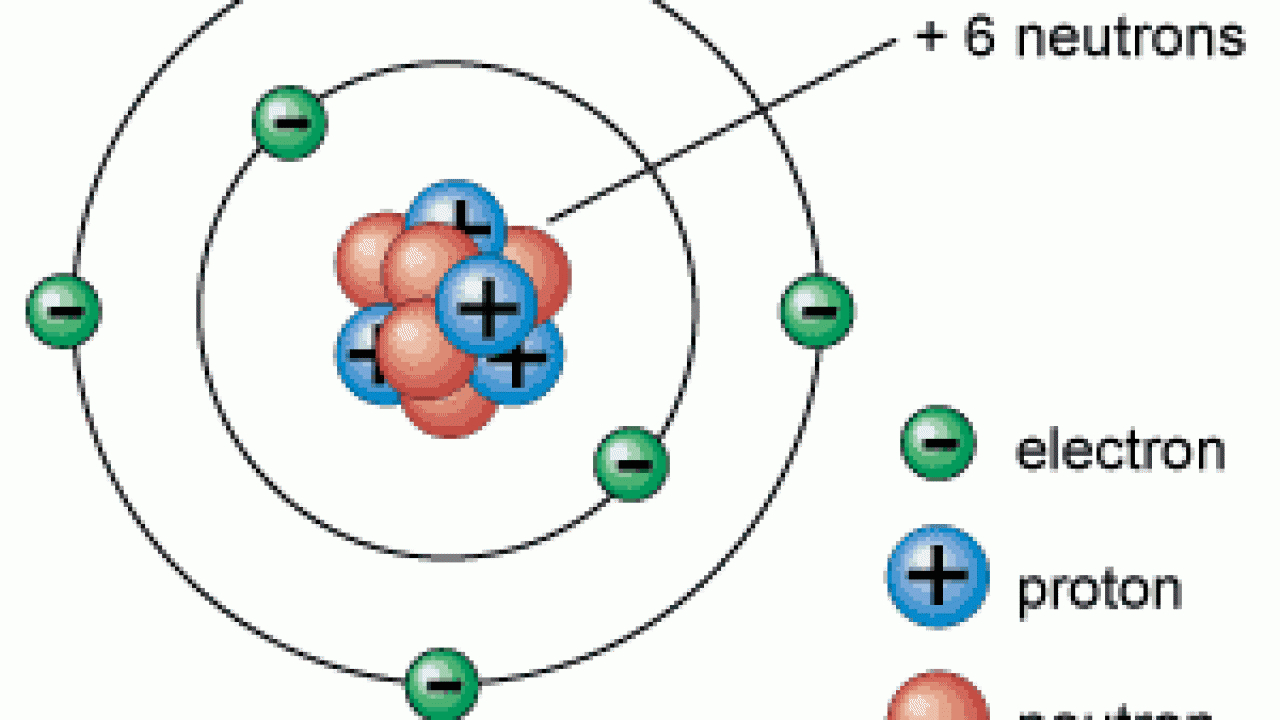
There is a maximum of seven shells, which can hold a different number of electrons, the electrons assignet to the outermost shell are known as valence electrons. Unlike the electron, the nucleus can be broken up into smaller building blocks called protons and neutrons. Neutrons have a neutral charge (no charge). Protons and neutrons are in the center of the atom, and they make up the nucleus.
Calculate the inner diameter of the tube in centimeters.

The mass of ethanol needed to fill the tube is found to be 45.23 g. The next heavier atom, the noble gas helium, consists of two electrons, two protons, and two neutrons.Īccording to the Bohr model of the atom the electrons are assigned to so-called shells, which represent different energy levels and therefore are arranged concentrically around the nucleus. Important concepts: Atoms consist of very small particles called protons, neutrons, and electrons the parts of the atom. A 25.0-cm-long cylindrical glass tube, sealed at one end, is filled with ethanol. The simplest atom is the hydrogen atom, with only one electron, one proton, and no neutron. We also learnt that electrons are negatively charged ( ) and revolve around the nucleus of the atom in concentric paths called orbits. The arrangement of electrons and protons are different in different elements. A naturally occurring atom is electrically neutral, there are just as many positive protons as negatively charged electrons inside an atom. We all studied Structure of an Atom in science that atoms are made up of 3 particles electrons, protons, and neutrons. The positively charged protons and the neutrons form the nucleus, which is encircled by the electrons in certain intervals. The number of protons in an atom determines. Neutrons, protons and electrons are the elementary particles. All other elements have the same basic structure: protons and neutrons at the center, and electrons orbiting around. Thomson was the first person who proposed a model for the structure of an atom. Thomson discovered the sub-atomic particle namely ‘electron.’. The atom of all elements (103 known) is composed of the smallest and elementary particles. The atomic number(Z) of an element is 11 and the atomic mass(A) is 23, a)Identify the element. By 1900, it was discovered that the atom was not a simple, indivisible particle, but rather it contains sub-atomic particles. 3.9) indicate each element has a distinctive number of protons and electrons. Depending on the atom, it is composed of a certain number of electrons, protons, and neutrons. The number of protons, neutrons, and electrons in an atom can be determined from a set of. The structure of atoms of some elements as shown (Fig. An atom is the smallest chemically not further divisible component of matter.
